Specific Gravity to Density Calculator
Turn specific gravity into density with a practical lab-style model. Enter SG, select a reference basis, add temperature and pressure inputs, and convert the result into kg/m3, g/mL, lb/gal, and lb/ft3.
| Water Temp C | Density kg/m3 | g/mL | lb/gal |
|---|---|---|---|
| 0 | 999.84 | 0.9998 | 8.345 |
| 4 | 999.97 | 1.0000 | 8.345 |
| 10 | 999.70 | 0.9997 | 8.343 |
| 20 | 998.21 | 0.9982 | 8.331 |
| 25 | 997.05 | 0.9971 | 8.321 |
| 40 | 992.22 | 0.9922 | 8.281 |
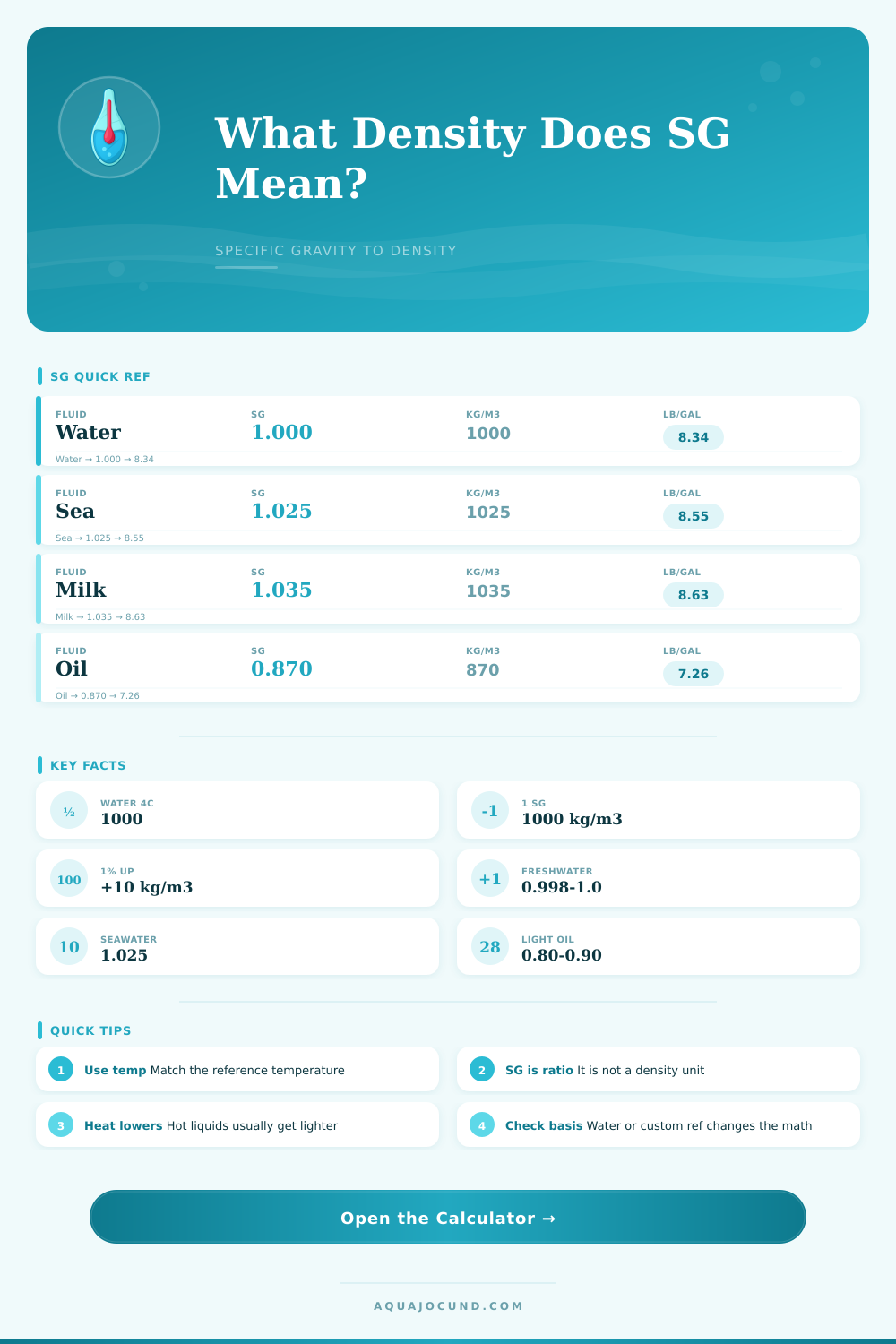
| Fluid | SG | Density kg/m3 | Note |
|---|---|---|---|
| Fresh water | 1.000 | 1000 | Baseline |
| Seawater | 1.026 | 1026 | Ocean mix |
| Milk | 1.035 | 1035 | Fat + solids |
| Motor oil | 0.870 | 870 | Floats |
| Glycerin | 1.260 | 1260 | Very dense |
| Corn syrup | 1.330 | 1330 | Thick mix |
| Unit | To Liter | To m3 | To gal |
|---|---|---|---|
| mL | 0.001 | 0.000001 | 0.000264 |
| L | 1 | 0.001 | 0.264172 |
| gal | 3.7854 | 0.003785 | 1 |
| ft3 | 28.3168 | 0.028317 | 7.48052 |
| Basis | Best For | Default Temp | Why Use It |
|---|---|---|---|
| Water at ref | Lab SG | Input temp | Most common |
| Water at 20 C | Shop work | 20 C | Easy compare |
| Custom | Process fluid | Any | Known density |
| Temp corrected | Hot fluids | User set | Less drift |
- Match temperatures: SG is most trustworthy when the sample and reference are close in temperature.
- Use a real baseline: Water at 20 C is a good default, but a measured custom density is better for process fluids.
- Check volume units: Mass from volume only makes sense when the unit conversion is set correctly.
- Round after you calculate: Keep extra precision through the math, then display your chosen decimals.
- Below 1.0: The liquid is lighter than water and usually floats.
- Above 1.0: The liquid is denser than water and tends to sink.
- Pressure is small: Most everyday liquid conversions barely change with pressure, but the option helps for consistency.
- Temperature matters: Hot liquids expand, so density drops unless the fluid is unusually structured.
Specific gravity are the ratio of the density of a substance to the density of a standard reference substance. The standard reference substance that many peoples use is water. People use water because it is a universal standard and allows people to understand if a liquid will float or sink.
A specific gravity reading of 1.000 mean that the density of the substance is the same than the reference water. When the reading of specific gravity is lower than 1.000, the substance is less dense than water, which means that it will float. For substances with a specific gravity reading that is higher than 1.000, the substance is more denser than water and will sink.
Specific Gravity and How Temperature Affects Density
To find the actual density of the substance, one can use the specific gravity reading. To find the actual density of a liquid, one must multiply the specific gravity of that liquid by the density of the water reference substance. However, the density of water change with the temperature of that water.
Therefore, it is necessary to account for the temperature of the water to ensure that the actual density calculation for the liquid is accurate. The temperature of the water has a critical impact on the density of the water and the substance being measure. Water reach its maximum density at around 4°C. Thus, as the temperature of the water increases, the density of the water decrease.
If the temperature of the water is not accounted for when calculating the density of the liquid, the density calculation will be incorrect. This incorrect calculation of density will create error in the pump settings of the liquid. Additionally, the liquids will also expand with an increase in temperature.
Thus, as the temperature of the liquid increases, the volume of that liquid increase. As a result, the density of that liquid will decrease. Some liquids will expand at a faster rate than other.
For instance, ethanol will expand at a faster rate than oils or glycerin. Thus, if the temperature of the liquid is not corrected for, the specific gravity reading for that hot liquid will be incorrect. This incorrect reading will also create errors in the pump that handle that liquid.
The reference choice for specific gravity readings are important. Many laboratories will use the temperature of the liquid as the reference temperature for the measurement of specific gravity. However, many shops will use 20°C as the reference temperature to make it easy for individuals to compare the specific gravity of liquids.
Additionally, individuals can set custom references for specific fluids to account for the known baseline density for those fluids. For instance, instead of calculating the specific gravity of a fluid relative to water at 20°C, the specific gravity could be calculate relative to the density of ethanol. Pressure will have a slight effect on specific gravity and density measurement for most ambient work environments.
There are errors that may occur in the measurement of specific gravity if the proper procedures are not follow. For instance, many people will often forget that the specific gravity only measure the weight of the substance under the influence of gravity, not the mass of that substance. Additionally, air buoyancy will affect the measurement of the mass of small sample of liquids.
Bubbles within the liquid will also skew the specific gravity measurement of the substance. The hydrometer that is used to measure the specific gravity must be clean and calibrated to ensure the specific gravity measurement of the liquid. Additionally, the scale on the hydrometer must match the reference temperature of the liquid being measured.
Otherwise, the ratio will be incorrect. Depending on the density of the substance being measured, it can be classified as a light, neutral, or heavy liquid. Liquids with a density that are below 900 kg/m³ are considered to be light liquids.
Light liquids, such as oils and solvents will float. A density of 1000 kg/m³ mean that the liquid has the same density as water and will have neutral buoyancy. Finally, liquids with a density above 1200 kg/m³ are considered heavy liquids, such as syrups and salts.
These types of liquid can settle within containers and may require agitation to prevent from settling. Additionally, the most heavy of the liquids, such as battery acid blends will require reinforced tank to contain the strong liquid. Knowing the density range of the liquids allows people to make decision regarding the pumps and seals that are used in the movement of these liquids.
There are also steps that can be taken to verify the results of the density measurement calculations. For instance, one can check the calculated density of the substance against the specification provided by the supplier sheets of the substance. Additionally, the gram per milliliter can be verified to be the same as the grams per cubic centimeter of the substance.
By using decimal in the calculations, it is possible to achieve high accuracy of the density calculation of the liquids. Finally, these decimal values can be rounded to present an accurate report of the density of the liquids. Thus, by following these procedure, individuals can turn the specific gravity of a substance into an accurate measurement of density.