🐟 Aquarium Ammonia Calculator
Calculate toxic free ammonia (NH3) vs safe ammonium (NH4+) based on pH, temperature, and TAN
| Level | NH3 Range (ppm) | Effect on Fish | Action Needed |
|---|---|---|---|
| Safe | < 0.02 | No harmful effects | Routine maintenance |
| Caution | 0.02 – 0.05 | Mild gill irritation, stress | 25–30% water change, check filter |
| Danger | 0.05 – 0.2 | Gill damage, gasping, lethargy | 50% water change immediately |
| Critical | 0.2 – 0.5 | Organ damage, fin rot, burns | 75% water change, add detoxifier |
| Lethal | > 0.5 | Rapid death likely within hours | Emergency 90% change, rehome fish |
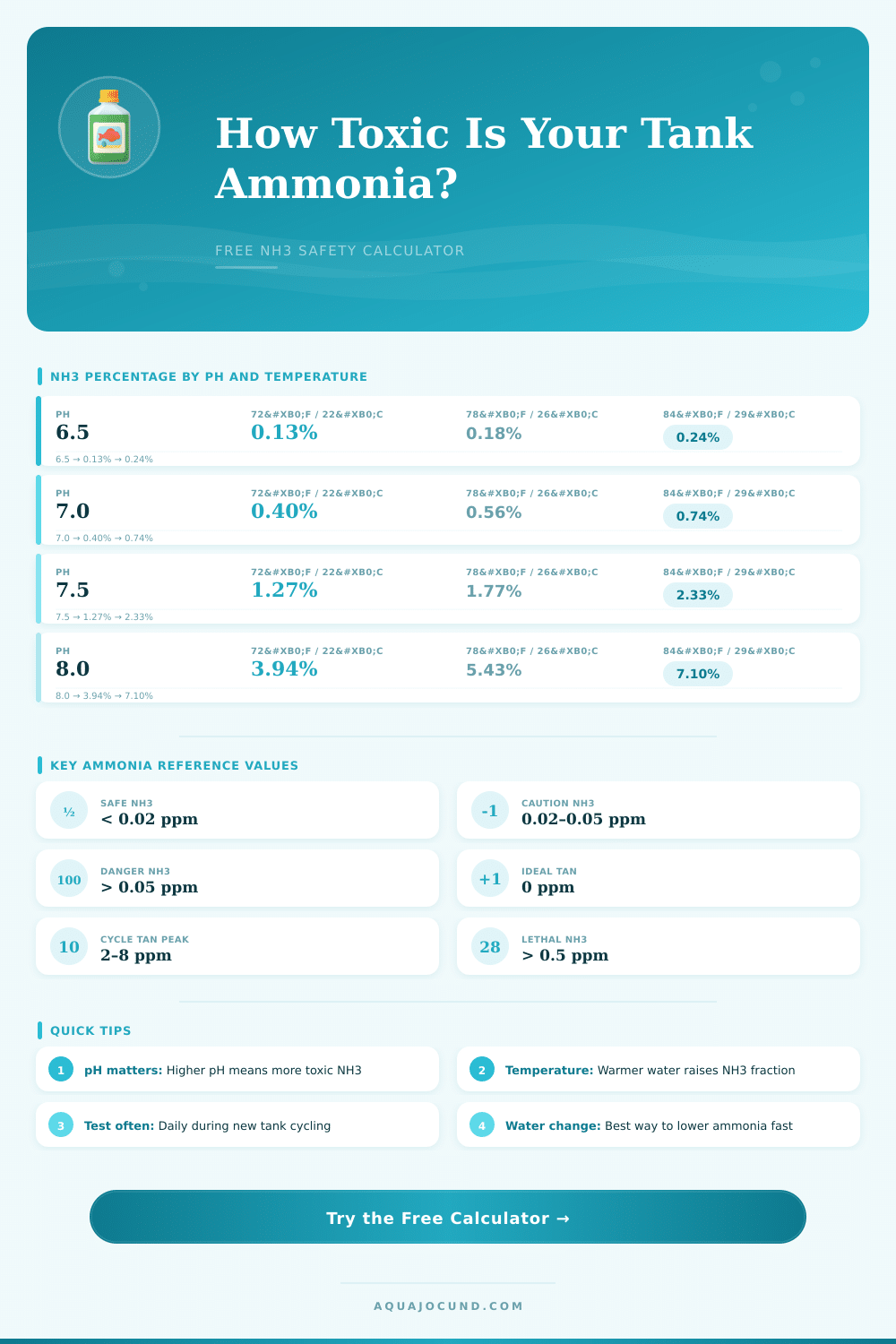
| pH | 64°F / 18°C | 72°F / 22°C | 78°F / 26°C | 84°F / 29°C |
|---|---|---|---|---|
| 6.0 | 0.01% | 0.02% | 0.02% | 0.03% |
| 6.5 | 0.04% | 0.05% | 0.07% | 0.09% |
| 7.0 | 0.13% | 0.18% | 0.24% | 0.30% |
| 7.5 | 0.40% | 0.56% | 0.74% | 0.94% |
| 8.0 | 1.27% | 1.77% | 2.33% | 2.93% |
| 8.5 | 3.94% | 5.43% | 7.10% | 8.82% |
| 9.0 | 11.68% | 15.66% | 19.82% | 23.97% |
| Scenario | Typical pH | Typical TAN | Notes |
|---|---|---|---|
| New tank cycling (day 7–14) | 7.2 – 7.8 | 2.0 – 8.0 ppm | Ammonia spikes expected; do not add fish |
| Established community tank | 6.8 – 7.4 | 0 – 0.25 ppm | Healthy biofilter keeps TAN near zero |
| Overstocked tank | 7.0 – 7.6 | 0.5 – 2.0 ppm | Biofilter overwhelmed; reduce stock |
| After overfeeding | 7.0 – 7.5 | 0.25 – 1.0 ppm | Remove uneaten food; skip next feed |
| African cichlid tank | 7.8 – 8.5 | 0 – 0.5 ppm | High pH makes even low TAN risky |
| Reef / marine tank | 8.1 – 8.4 | 0 – 0.1 ppm | Must be undetectable for corals |
| Discus tank (soft water) | 5.5 – 6.5 | 0 – 0.5 ppm | Low pH converts most TAN to safe NH4+ |
| Shrimp breeding tank | 6.5 – 7.0 | 0 – 0.25 ppm | Shrimp are very sensitive to NH3 |
Ammonia in its pure form, that is NH₃, truly harms fish, even in very small amounts. Each Aquarium keeper should aim for Ammonia levels that stay almost at zero and ideally they stay there unchanged. In water, where fish live, the Ammonia reading should show 0 ppm.
If it goes past that, then you must right away find the cause.
Ammonia in Aquariums: Danger, Causes and How to Fix It
Here it becomes more interesting. Most of the Ammonia that you find in an Aquarium is not pure Ammonia. It is ammonium, or ionised Ammonia (NH₄⁺).
That mostly does not matter for your fish. So some of that ammonium changes to pure Ammonia (NH₃), and here starts real trouble. The dfference between those two forms depends fully on pH and temperature.
One can have very high total Ammonia readings without enough pure Ammonia to cause real damage.
What so puts all this Ammonia in your Aquarium at first? Fish give it off all the time. Through their gills, urine and waste, they simply breathe it out daily.
They are the main source, simply said. Uneaten food and rotting matter on the bottom also add too the problem. One cool fact: animals like shrimps and snails do not make Ammonia like fish do.
In a healthy Aquarium, helpful bacteria break Ammonia into less harmful forms. Those Ammonia-eating bacteria do the main work. They change Ammonia first into nitrites, later other bacteria take the nitrites and turn them into nitrates.
When the nitrogen cycle fully sets itself up, the Aquarium is considered cycled, and Ammonia should stay at zero.
Many fishkeepers cycle their tanks without fish, using pure Ammonia for food. Five doses of pure Ammonia daily can start the process well. When nitrites appear in your tests, you lower the Ammonia dose.
While cycling, Ammonia at 1 to 2 ppm seems to work most well. Bottled bacteria products sometimes help speed things up. Adding material from an old, stable Aquarium or moving an old filter into the new setup also starts helpful bacteria colonies well.
Make sure that Ammonia reaches zero before adding fish.
If Ammonia sharply rises in a stable Aquarium, water changes are the fastest help. Remove 20 to 50 percent of the water and replace with fresh, treated water, that matches the tank temperature. Daily changes of 25 to 40 percent help to control Ammonia, while bacteria catch up and grow.
During cleaning of filter material, wash it in Aquarium water, not in tap water, to protect the helpful bacteria that you built with effort.
Tap water itself sometimes holds Ammonia in your system. Some water companies use chloramine, that splits and releases Ammonia as a side effect. That issurprising, and nobody wants that, especially if the water company did not warn about the change.